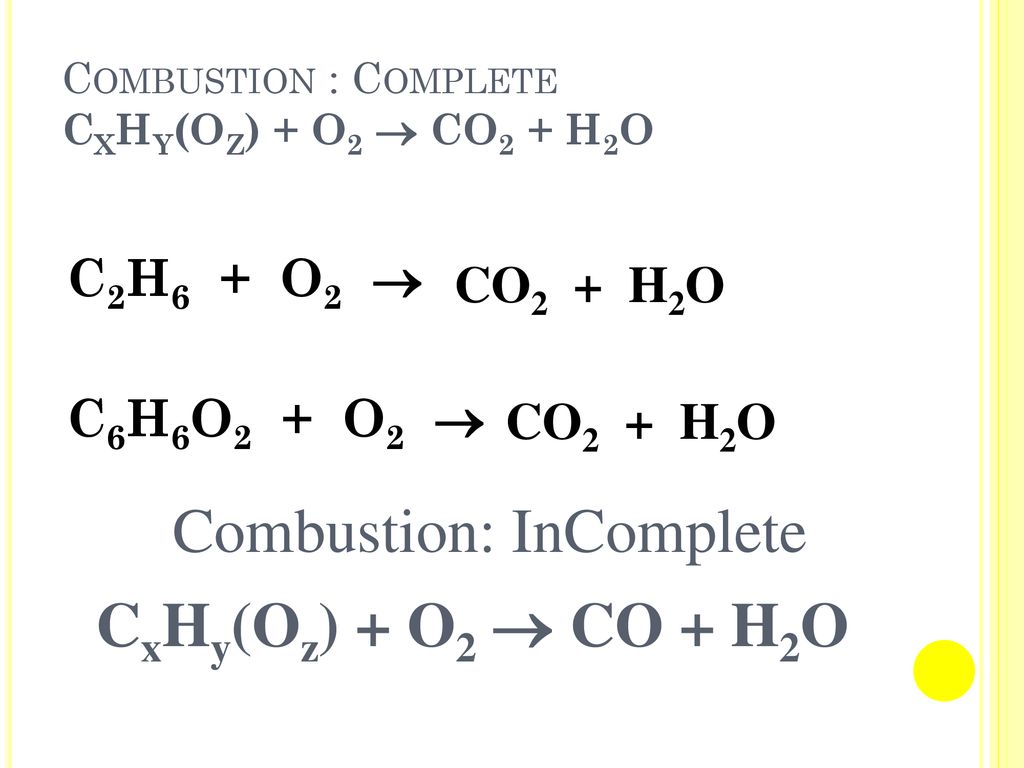Drill #4B 2/10/15 Predict the products and balance the equation for the following double-replacement reaction: Na2CrO4 (aq) + Ba(OH)2 (aq) - ppt download

SOLVED: Balance the equation by inserting coefficients as needed: equation: C6H6 + O2 -> CO2 + H2O Classify the reaction: combustion

PPT - Chemical Equations reactants products reactant - starting substance in a chemical reaction PowerPoint Presentation - ID:5812787
Balance the following equation by oxidation number method. C6H6 + O2 → CO2 + H2 O - Sarthaks eConnect | Largest Online Education Community
how to balance chemical equation c6h6+o2 arrow mark co2+h20 but. i tried and in RHS why should we put 12 next to carbon instead of that we can put put 3 so

Balance this equation by oxidation number method (iv) C6H6 + O2 → CO2 + - Chemistry - Redox Reactions - 11918394 | Meritnation.com

C6H6 +O2 ------}CO2 + H2O balance By oxi Methods - Chemistry - Redox Reactions - 13233357 | Meritnation.com

C6H6+O2(g)→CO2(g)+H2O(g) When the equation the reaction represented above is balanced and all coefficients are reduced to lowest whole number terms, the coefficient H2O(g) is :








![Tamil] Balance the following equation by oxidation number method Tamil] Balance the following equation by oxidation number method](https://d10lpgp6xz60nq.cloudfront.net/physics_images/FM_CHE_XI_V01_C01_E04_188_S01.png)